TECHNICAL SPECIFICATIONS
| Item | Specification |
|---|---|
| Product Name | Mono(6-amino-6-deoxy)-β-cyclodextrin |
| CAS NO. | 29390-67-8 |
| Appearance | White to off-white crystalline powder |
| Molecular Formula | C42H71NO34 |
| Molecular Weight | 1134.00 g/mol |
| Purity (RP-HPLC) | ≥98.0% |
| Solubility | Freely soluble in water, DMSO, DMF; slightly soluble in methanol |
| Endotoxin | ≤10 EU/mg |
| Biological Activity | Effective complexation with hydrophobic molecules; enhances solubility of poorly water-soluble drugs |
| Storage Conditions | Store at 2–8°C, protected from light and moisture |
| Stock Location | Stock in USA |
| MOQ | 1g |
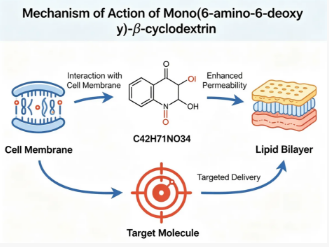
1.MECHANISM OF ACTION
The primary mechanism of action revolves around the dual functionality of the molecule: the hydrophobic inner cavity provides a microenvironment to accommodate guest molecules through non-covalent interactions, while the exposed amino group at the rim offers a site for protonation in neutral to acidic pH conditions, facilitating electrostatic interactions with anionic compounds. Additionally, this primary amine acts as a robust chemical handle that enables covalent linkage to carboxylic acids, active esters, aldehydes, and other electrophiles under mild conditions, allowing for the rational design of complex architectures such as polymer conjugates, surface-immobilized sensors, and targeted nanocarriers. This combination of host-guest complexation and straightforward derivatization makes it a dynamic tool for modulating the physicochemical properties of bioactive agents and constructing responsive delivery vehicles
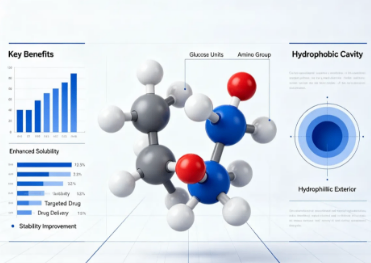
2.PRODUCT INDICATIONS
This product is indicated primarily for research and development purposes within pharmaceutical, biochemical, and material science sectors, targeting scientists and formulators working on drug solubilization, stabilization, and novel delivery platforms. It is particularly suited for professionals developing inclusion complexes to enhance the bioavailability of hydrophobic drugs, as well as researchers designing functionalized cyclodextrins for use in catalysis, chiral separation, and the fabrication of biosensors or diagnostic probes. Furthermore, it serves as a critical reagent for synthetic chemists aiming to introduce cyclodextrin moieties into larger molecular frameworks or surfaces, thereby catering to the needs of advanced material engineers and bioconjugation specialists.
3.CLINICAL EFFICACY OF PRODUCT
As a chemical reagent and research intermediate, Mono(6-amino-6-deoxy)-β-cyclodextrin does not possess direct clinical efficacy in the therapeutic sense; rather, its value is demonstrated through its performance in preclinical formulations and experimental setups where it significantly improves the solubility, stability, and controlled release profiles of associated active pharmaceutical ingredients. Studies and formulation trials indicate that derivatives and complexes formed using this amino-functionalized cyclodextrin can enhance drug loading capacity and modify pharmacokinetics, paving the way for more effective delivery systems, though the product itself is strictly intended for laboratory use and not for direct administration to humans or animals.
4.Certificate
GlobalTech Co., Ltd. is committed to providing high-purity biotechnological peptide products, adhering strictly to quality standards and obtaining internationally recognized certifications.
Our production partners all employ GMP-compliant production systems and possess standardized quality control processes covering raw material procurement, synthesis, purification, testing, packaging, and storage. Each batch of product comes with complete documentation, including a Certificate of Analysis (COA), HPLC purity report, mass spectrometry (MS) chromatograms, and third-party testing reports available upon request.
We prioritize:
1.A rigorous quality management system
2.Batch traceability and stability data
3.Professional laboratory testing and validation
4.Compliance with international trade standards
Our commitment to quality, transparency, and regulatory compliance ensures the provision of reliable, stable, and research-grade peptide products to our global partners.

5.DELIVERY & SHIPPING
We specialize in global logistics for various cosmetic peptides. Your order will be handled with the utmost care, ensuring safe, reliable, and intact delivery.
Professional and Secure Packaging
Each bottle of peptide is protected with a special packaging solution to ensure stability and integrity during transportation.
We can also customize packaging according to customer needs.
Shipping Methods
Main Method: International Air Freight
We primarily use DHL, FedEx, UPS, and TNT because of their global reliability, speed, and advanced tracking systems. Air freight ensures the fastest transit time and minimizes the impact of environmental factors on the product.

Global Technology Co., Ltd. is a modern and advanced enterprise specializing in the production and export of active pharmaceutical ingredients (APIs), peptides, and plant extracts. The company invests in and collaborates closely with domestic manufacturers holding GMP, DMF, and FDA certifications, as well as laboratories at renowned universities, to meet diverse customer needs ranging from gram-level to ton-level. This enables Dejurui Pharmaceutical to provide customers with more flexible and convenient access to cost-effective, high-quality products and superior services.
We focus on the research and development and supply of high-quality chemical products, strictly adhering to international standards and quality systems. Our products are stable in performance and reliable in purity, widely used in industrial manufacturing, scientific research, and the fine chemical industry. We provide solid support at every stage with a professional and responsible attitude.
Hot Tags: Loureirin D, Dragon's Blood D, Dihydrochalcone, P2Y12 Receptor Antagonist, Antiplatelet Agent, Natural Product, Pharmaceutical Intermediate, Research Chemical, Antithrombotic, Anti-inflammatory, Anti-fibrotic, NF-κB Inhibitor, Dracaena cochinchinensis Extract, Standard Reference Material, Biomedical Research.
If you have any product requirements, please send an email to:sales@huanqiukeji9.com;whatsapp:85244219157








![Ac-[DTrp16] Endothelin-1 (16-21), human CAS NO.143037-33-6 Ac-[DTrp16] Endothelin-1 (16-21), human CAS NO.143037-33-6](https://images.cnmqh.com/images/a10134/a13725cd07d2c2b755c5320f53d74e81-20260507065809.webp)




