TECHNICAL SPECIFICATIONS
| Item | Specification |
|---|---|
| Product Name | Icaritin |
| CAS NO. | 118525-40-9 |
| Appearance | Light yellow to off-white crystalline powder |
| Molecular Formula | C21H20O6 |
| Molecular Weight | 368.38 g/mol |
| Purity (RP-HPLC) | ≥98.0% |
| Solubility | Soluble in DMSO, methanol; slightly soluble in water |
| Endotoxin | ≤10 EU/mg |
| Biological Activity | Anti-tumor, anti-inflammatory, osteogenic, immunomodulatory |
| Storage Conditions | Store at -20°C, protected from light and moisture |
| Stock Location | Stock in USA |
| MOQ | 1g |
1.MECHANISM OF ACTION
The primary mechanism of action of Icaritin involves the multi-target regulation of critical signaling cascades, most notably the suppression of the IL-6/JAK2/STAT3 pathway which is hyperactivated in various tumors and inflammatory conditions. By interfering with the MyD88/IKKα protein complex, it effectively reduces the phosphorylation of STAT3, thereby inhibiting the expression of downstream pro-inflammatory cytokines such as TNF-α and IL-6, as well as immune checkpoint molecules like PD-L1. Furthermore, it modulates the tumor immune microenvironment by restricting the generation and suppressive function of myeloid-derived suppressor cells (MDSCs), which collectively restores the effector function of T cells and shifts the immune landscape from a tolerant to an active state against pathological cells.
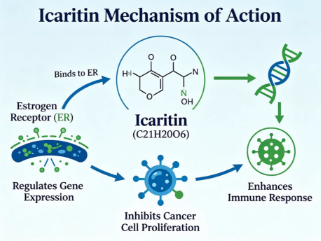
2.PRODUCT INDICATIONS
This product is indicated strictly for laboratory research, preclinical formulation, and analytical standard applications within pharmaceutical, nutraceutical, and cosmetic R&D sectors. It is particularly suited for researchers investigating novel anti-hepatocellular carcinoma (HCC) agents, scientists exploring natural phytoestrogens for bone regeneration and osteoporosis treatment, and formulators working on stabilizing poorly soluble botanical extracts. Additionally, it serves as a vital reference standard for analytical method development (such as HPLC calibration) and as a lead compound for medicinal chemists aiming to synthesize more bioavailable derivatives or novel anti-cancer drug candidates.
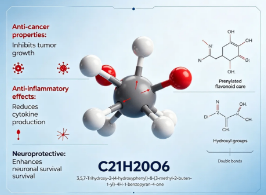
3.CLINICAL EFFICACY OF PRODUCT
While Icaritin as a raw material compound does not possess direct clinical efficacy for patient administration, its formulated pharmaceutical version (Icaritin soft capsules, branded as Acoradine) has demonstrated significant clinical benefits in approved therapies for advanced hepatocellular carcinoma. Clinical trial data highlights its ability to improve overall survival and progression-free survival in patients unsuitable for standard systemic therapies, particularly those with specific biomarker profiles, while maintaining a markedly superior safety and tolerability profile with fewer severe adverse events compared to traditional multi-kinase inhibitors. These clinical outcomes validate the compound's potent anti-tumor and immunomodulatory potential, making the high-grade research chemical indispensable for ongoing translational medicine studies.
GlobalTech Co., Ltd. is committed to providing high-purity biotechnological peptide products, adhering strictly to quality standards and obtaining internationally recognized certifications.
Our production partners all employ GMP-compliant production systems and possess standardized quality control processes covering raw material procurement, synthesis, purification, testing, packaging, and storage. Each batch of product comes with complete documentation, including a Certificate of Analysis (COA), HPLC purity report, mass spectrometry (MS) chromatograms, and third-party testing reports available upon request.
We prioritize:
1.A rigorous quality management system
2.Batch traceability and stability data
3.Professional laboratory testing and validation
4.Compliance with international trade standards
Our commitment to quality, transparency, and regulatory compliance ensures the provision of reliable, stable, and research-grade peptide products to our global partners.

5.DELIVERY & SHIPPING
We specialize in global logistics for various cosmetic peptides. Your order will be handled with the utmost care, ensuring safe, reliable, and intact delivery.
Professional and Secure Packaging
Each bottle of peptide is protected with a special packaging solution to ensure stability and integrity during transportation.
We can also customize packaging according to customer needs.
Shipping Methods
Main Method: International Air Freight
We primarily use DHL, FedEx, UPS, and TNT because of their global reliability, speed, and advanced tracking systems. Air freight ensures the fastest transit time and minimizes the impact of environmental factors on the product.

Global Technology Co., Ltd. is a modern and advanced enterprise specializing in the production and export of active pharmaceutical ingredients (APIs), peptides, and plant extracts. The company invests in and collaborates closely with domestic manufacturers holding GMP, DMF, and FDA certifications, as well as laboratories at renowned universities, to meet diverse customer needs ranging from gram-level to ton-level. This enables Dejurui Pharmaceutical to provide customers with more flexible and convenient access to cost-effective, high-quality products and superior services.
We focus on the research and development and supply of high-quality chemical products, strictly adhering to international standards and quality systems. Our products are stable in performance and reliable in purity, widely used in industrial manufacturing, scientific research, and the fine chemical industry. We provide solid support at every stage with a professional and responsible attitude.
Hot Tags: Loureirin C, Dragon's Blood C, Dihydrochalcone, NRF2 Activator, Antioxidant, Neuroprotective Agent, Thrombin Inhibitor, Natural Product, Research Chemical, Anti-ferroptosis
If you have any product requirements, please send an email to:sales@huanqiukeji9.com;whatsapp:85244219157








![Ac-[DTrp16] Endothelin-1 (16-21), human CAS NO.143037-33-6 Ac-[DTrp16] Endothelin-1 (16-21), human CAS NO.143037-33-6](https://images.cnmqh.com/images/a10134/a13725cd07d2c2b755c5320f53d74e81-20260507065809.webp)




